Contract Research Organisations (CROs)
Ladda ner PDF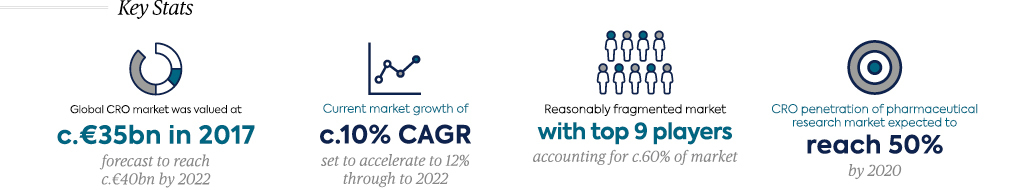
CROs have evolved to become strategic partners of their sponsors/pharma companies and high-growth of the market is the result of the following factors:
The need to reduce R&D expenditure
With rising costs of R&D, it is becoming increasingly inefficient for innovators to maintain development teams and facilities. Outsourcing has allowed innovators to rationalise fixed R&D cost bases and achieve operational efficiencies.
Increasing complexity of drugs and clinical trials
The complexity of therapeutics in development is constantly increasing, making them more costly and challenging to develop in-house. Furthermore, heightened regulatory protocols have increased the duration, cost and complexity of clinical trials.
Time and efficiency
Innovators are increasingly reliant on CROs to optimise R&D activities, shorten development timelines, reduce attrition rates and expand clinical trial management capabilities globally.
Expanding CRO solutions and technologies
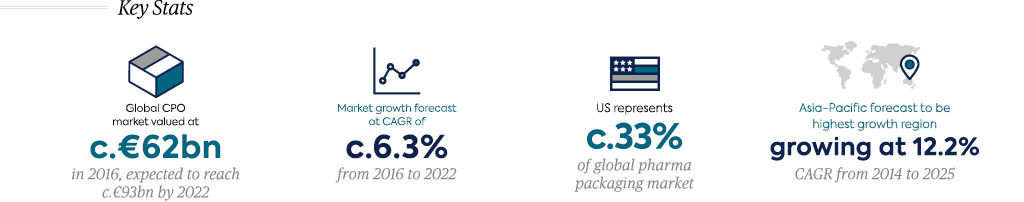
CROs are constantly expanding their addressable markets by broadening their service offerings, capturing an increasing share of pharma R&D expenditure. Investment in technology and big data will continue to play an important role in the expansion of the CRO sub-sector. As a result, CRO penetration has steadily grown, accounting for c.34% of biopharma clinical development spend in 2011 and expected to increase to c.50% by 20207 (valuing the global clinical CRO market at €13.7bn in 20207). Despite the growing range of services offered by CROs, they can broadly be grouped into the three following categories:
- Pre-clinical
Pre-clinical services can be sub-divided into drug discovery and pre-clinical trial services. Drug discovery services are typically lab-based and involve the identification of promising ‘lead compounds’. Once selected for further research, the molecules then enter preclinical trials. Assuming the compound shows signs of efficacy and is safe, the innovator will submit an Investigational New Drug Application (IND). If approved by regulators, the innovator has permission to proceed with clinical trials.
- Clincal
Clinical trials are typically conducted inthree distinct Phases (I, II and III), each with different objectives, increasing numbers of patients and costs.- Phase I trials are focused on basic safety and pharmacology in patients who may not necessarily have the target disease (c.20-100 patients). These studies are typically conducted at specialised research centres and are designed to monitor the metabolic reactions and patient tolerance to the compounds at multiple dosage ranges.
- Phase II and III trials are primarily efficacy studies on patients afflicted by the target disease. Phase II trials (often known as proof of concept trials) test efficacy alongside dose ranging and further safety testing (c.100-500 patients). Phase III trials are much larger (c.500-1000 patients), in which advanced efficacy and safety testing are conducted at multiple testing centres. These are typically the longest and most expensive trials, and regulatory authorities typically require two successful Phase III trials to obtain approval.
- Post Approval
Post-approval, regulatory agencies typically require innovators to collect and periodically report additional safety and efficacy data (sometimes referred to as real world evidence or ‘RWE’ studies). If marketed internationally then surveillance data from all countries must be collected. Across clinical and post-approval trials, many CROs have developed a full suite of services allowing innovators to fully outsource their R&D activities and partner with CROs in the design and delivery of research operations. Services CROs typically offer include trial planning, project management, patient recruitment, site access, clinical staffing, patient monitoring, pharmacovigilance and data analysis.
The market remains reasonably fragmented, with the top nine players accounting for c.60% of the c.€35bn global market7 and several hundred smaller players making up the remainder of the market. The market has undergone a period of intense consolidation over the last decade as a result of numerous landmark mergers (e.g. LabCorp/Chiltern, INC/ InVentiv, LabCorp/Covance), primarily driven by the desire of Big Pharma to partner with fewer full-service providers. Despite this, among small and midsized players, there is often a preference to partner with mid-sized CROs that will focus on maintaining a long-term quality relationship. Small and mid-sized biopharmaceutical companies are also more likely to partner with CROs with a therapeutic focus or specific expertise.