Contract Commercialisation Market (CCOs)
Download PDF
CCOs offer a broad range of commercialisation services to their customers, typically focussed on preparing new products for launch and maximising sales post approval.
CCO services can broadly be divided into four sub-sectors:
Medical affairs
Services dedicated to communicating the clinical rationale, evidence and best practice for a therapy to healthcare professionals (HCPs).
Marketing
Focussed on activities relating to establishing a successful brand, including analysis of the target market, developing a brand proposition and engagement strategies for key stakeholders.
Market access
Activities relating to making a drug available to patients, including developing pricing and reimbursement strategies, healthcare economics and outcomes research (HEOR) to support pricing strategies and payer/KOL engagement services.
Other
The CCO sector has been particularly resilient to COVID-19
Predominantly regulatory, technical and operational services, such as pharmacovigilance, to support the commercialisation of a product post launch.
Despite the high levels of recent M&A activity in the sector, the less mature nature of the CCO market means that it remains highly fragmented, with players ranging from small agencies or consultancies specialising in specific services or regions to large, diversified CCOs with global operations.
Whilst medicines can have a lifecycle of 20+ years with outsourced commercialisation services required throughout this lifecycle, the majority of this spend is focussed immediately prior to and in the years immediately following launch, although the distribution of this spend varies by sub-sector according to the nature of the services in relation to the drug development cycle.
Medical affairs
Expenditure usually commences around Phase II, following which spend increases until its peak during launch, in line with HCP engagement activity both pre and post launch.
Marketing
Marketing is the largest sub-sector, with spend typically beginning around phase III and peaking during the launch phase as promotional activities intensify.
Market access
Spend is typically the earliest in the drug lifecycle, given the need to assess the likely pricing and reimbursement landscape and the commercial prospects of a drug early in its development.
Other
Other CCO services are typically regulatory or operational in nature and therefore, spend on these tend to be later in the drug development and sustained post the launch phase.
Given the increasing cost and complexity of R&D, successful execution of commercial strategies is becoming increasingly important, and biopharma are investing in commercialisation services earlier in the drug lifecycle as a result, across all sub-sectors.
COVID-19 impacts on the CCO sub-sector
With the exception of field sales services, the CCO sector has been particularly resilient to COVID-19, with a majority of companies in the sector being virtual organisations performing desktop-based work for global biopharma customers. As such, most of these service providers have transitioned well to 'working from home' environments, with minimal COVID-19 related disruption. The resilience of the CCO market combined with its fragmented nature has resulted in significant levels of M&A activity since the onset of the pandemic, a majority of it driven by PE.
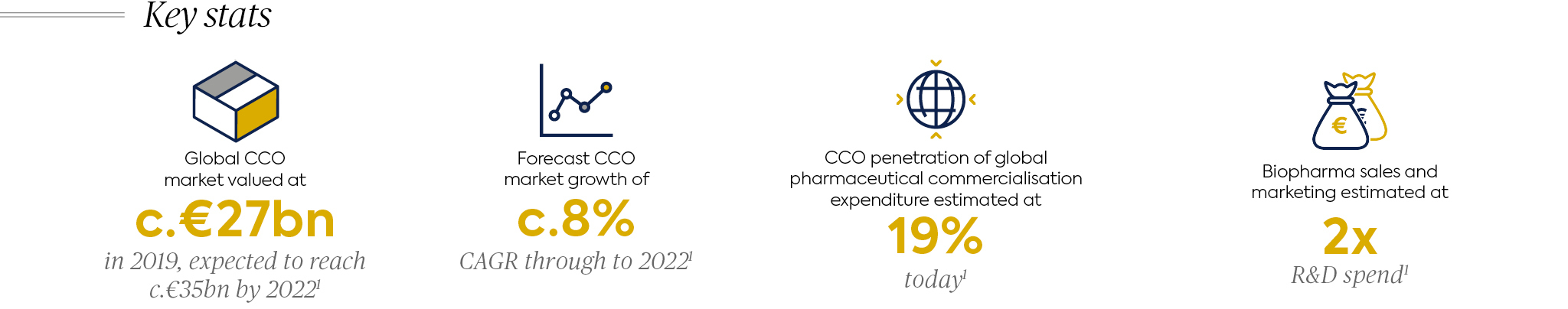
CCO M&A trends
Platform expansion
Recent M&A activity in the sub-sector has been driven by the desire to consolidate but also to diversify, driven by pharma’s wish to work with fewer outsourced service providers. Although much of this has been driven by PE bolt-on M&A activity, trade acquirers are also seeking to diversify their service portfolios via M&A. A good case study of this is Ashfield (UDG) who have acquired the following new capabilities through M&A, in order to create a diversified network of services to its global customer base:
- Market access, through its acquisition of PHMR in 2021 for €38m.
- Strategic consultancy, through its acquisition of Putnam Associates in 2019 for €79m.
- Medical communications, through its acquisition of Create NYC in 2018 for €59m.
Large players consolidating CRO and CCO operations
A number of the larger CRO providers have achieved substantial synergies by adding commercialisation services to their portfolios through M&A, allowing them to provide an ‘end-to-end’ solution to their clients throughout the drug life cycle. Example of such transactions include the INC/Inventiv merger (creating Syneos), the Quintiles/ IMS merger (creating IQVIA) and PPD’s acquisition of Evidera.
PE exits in the sector have also increased the attractiveness of the sector to many generalist funds
Increasing importance of data and technology
The use of data and technology has revolutionised the CCO market, accelerated during the COVID-19 pandemic. A number of recent deals at high valuations have underlined the importance the market places on having access to data and technologies which can provide a competitive advantage over peers. Recent examples of such transactions include Clarivate Analytics’ recent acquisition of DRG for €863m, GHO’s recapitalisation of Envision Pharma (both 2020), and
New Mountain Capital’s investment in W2O in 2019.
High levels of PE activity in the sector
PE activity in the sector has accelerated throughout COVID-19, primarily driven by the high growth and resilient nature of the sector, which has faced relatively little disruption during the pandemic. Recent successful PE exits in the sector have also increased the attractiveness of the sector to many generalist funds, increasing both the competitive tension and valuations in M&A processes. In addition to those noted above, recent notable PE deals in the sector include, ICGs acquisition of Lucid in 2021, Northedge’s acquisition of Helios in 2021, Bridgepoint’s acquisition of Prescient in 2021 and Fishawack in 2020, and Waterland’s acquisition of IMC in 2020.












